R. Sevilla, H. Gao, K. J. Steinberg, and B. M. Gallant
Elucidating Concentration-Dependent Energy Limitations in Li Primary Battery Fluoro-organosulfur Catholytes
Journal of Physical Chemistry C, 2023, DOI: 10.1021/acs.jpcc.2c07659
[publisher link]
Abstract
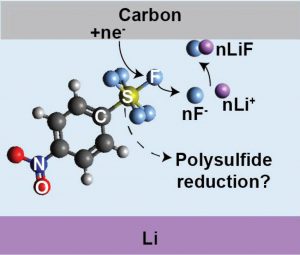
Unlocking longer-lasting lithium (Li) primary batteries that exceed the energy density of leading Li–carbon monofluoride (Li–CFx) requires exploration of new cathode redox processes. It was recently reported that a Li–carbon cell containing a liquid cathode + electrolyte (catholyte) based on the reactant 4-nitrophenylsulfur pentafluoride (NO2-Ph-SF5) can achieve competitive energy density (1085 Wh kg–1) as Li–CFx (∼1000 Wh kg–1), normalized to the weight of electrodes + catholyte or electrolyte in each respective cell. However, at high reactant concentrations (4–5 M) necessary to maximize total energy, the attained capacities fall significantly below the theoretical value. Here, we investigated the root causes of this shortfall by scrutinizing the critical role of NO2-Ph-SF5 concentration on reactant utilization and discharge intermediates/products. Despite the generation of electronically insulating lithium fluoride (LiF) throughout discharge, the capacity at high concentrations (4 M) is not limited by carbon passivation, but rather by the catholyte’s inability to solubilize and retain activity of key polysulfide-like intermediates. By examining a series of catholyte cosolvents in detail, we further identify solvent properties that support maximal capacities. These findings point toward future strategies to improve utilization of fluoro-organosulfur catholytes by simultaneously designing for high solubility of discharge products (LiF) and intermediates (S-based species), which requires looking beyond conventional solvents.

Recent Comments